"Standard atomic weights of the elements 2021 (IUPAC Technical Report)". ^ Prohaska, Thomas Irrgeher, Johanna Benefield, Jacqueline et al."The NUBASE2020 evaluation of nuclear properties" (PDF). ^ a b c d e Theoretically capable of spontaneous fission.^ Believed to undergo β +β + decay to 132Xe with a half-life over 300×10 18 years.^ Cluster decay is predicted but had never been observed.^ # – Values marked # are not purely derived from experimental data, but at least partly from trends of neighboring nuclides (TNN).^ ( ) spin value – Indicates spin with weak assignment arguments.^ Bold symbol as daughter – Daughter product is stable.^ Bold italics symbol as daughter – Daughter product is nearly stable.^ # – Atomic mass marked #: value and uncertainty derived not from purely experimental data, but at least partly from trends from the Mass Surface (TMS). 
^ ( ) – Uncertainty (1 σ) is given in concise form in parentheses after the corresponding last digits.However this decay is not yet observed the upper limit on the branching ratio of such decay is 0.0034%. The shorter-lived 137mBa (half-life 2.55 minutes) arises as the decay product of the common fission product caesium-137.īarium-114 is predicted to undergo cluster decay, emitting a nucleus of stable 12C to produce 102Sn. The longest-lived isomer is 133mBa, which has a half-life of 38.9 hours. All other radioisotopes have half-lives shorter than two weeks. The longest-lived of these is 133Ba, which has a half-life of 10.51 years. 
There are a total of thirty-three known radioisotopes in addition to 130Ba. This nuclide decays by double electron capture (absorbing two electrons and emitting two neutrinos), with a half-life of (0.5–2.7)×10 21 years (about 10 11 times the age of the universe). The Standard English unit is pounds mass per cubic foot ( lbm/ft 3).īarium – Properties Summary Element Barium Atomic Number 56 Symbol Ba Element Category Alkaline Earth Metal Phase at STP Solid Atomic Mass 137.327 Density at STP 3.51 Electron Configuration 6s2 Possible Oxidation States +2 Electron Affinity 13.95 Electronegativity 0.89 1st Ionization Energy 5.2117 Year of Discovery 1808 Discoverer Davy, Sir Humphry Thermal properties Melting Point 725 Boiling Point 1640 Thermal Conductivity 18 Specific Heat 0.204 Heat of Fusion 7.Naturally occurring barium ( 56Ba) is a mix of six stable isotopes and one very long-lived radioactive primordial isotope, barium-130, identified as being unstable by geochemical means (from analysis of the presence of its daughter xenon-130 in rocks) in 2001. The standard SI unit is kilograms per cubic meter ( kg/m 3). In other words, the density (ρ) of a substance is the total mass (m) of that substance divided by the total volume (V) occupied by that substance. It is an intensive property, which is mathematically defined as mass divided by volume: Typical densities of various substances at atmospheric pressure.ĭensity is defined as the mass per unit volume. How does the atomic mass determine the density of materials? Density of Barium The atomic mass number determines especially the atomic mass of atoms. The mass number is different for each different isotope of a chemical element. For 63Cu, the atomic mass is less than 63, so this must be the dominant factor. A nucleus with greater binding energy has lower total energy, and therefore a lower mass according to Einstein’s mass-energy equivalence relation E = mc 2. The nuclear binding energy varies between nuclei.This increases the mass of nuclei with more neutrons than protons relative to the atomic mass unit scale based on 12C with equal numbers of protons and neutrons. The neutron is slightly heavier than the proton.There are two reasons for the difference between mass number and isotopic mass, known as the mass defect: For example, 63Cu (29 protons and 34 neutrons) has a mass number of 63, and an isotopic mass in its nuclear ground state is 62.91367 u. For other isotopes, the isotopic mass usually differs and is usually within 0.1 u of the mass number. One unified atomic mass unit is approximately the mass of one nucleon (either a single proton or neutron) and is numerically equivalent to 1 g/mol.įor 12C, the atomic mass is exactly 12u, since the atomic mass unit is defined from it. One atomic mass unit is equal to 1.66 x 10 -24 grams. The unit of measure for mass is the atomic mass unit (amu). Therefore this resulting atomic mass is calculated from naturally-occurring isotopes and their abundance. 
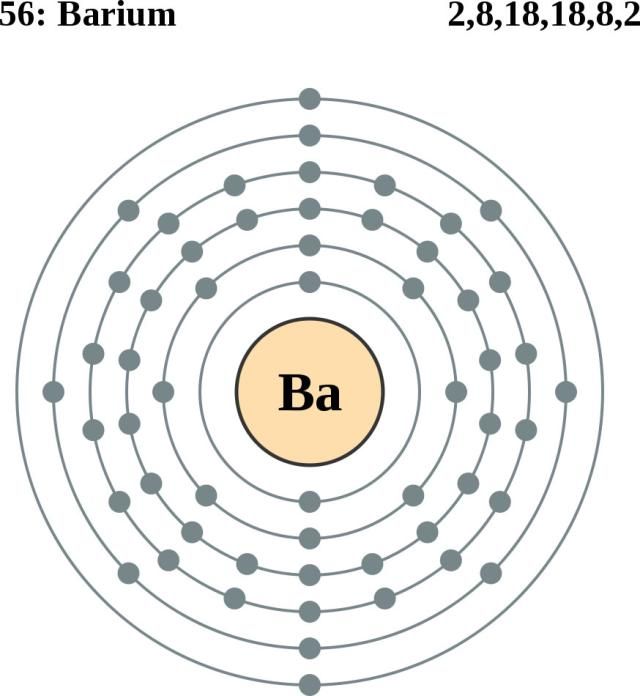
Note that each element may contain more isotopes. How does the atomic number determine the chemical behavior of atoms? Atomic Mass of Barium Since the number of electrons is responsible for the chemical behavior of atoms, the atomic number identifies the various chemical elements. Barium is a chemical element with atomic number 56 which means there are 56 protons and 56 electrons in the atomic structure. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
